Diagnosis of Feline Respiratory Mycoplasma Infection

Bioguard Corporation In cats, ’mucosal’ mycoplasma infections typically cause ocular and respiratory disease, and less frequently neurological or joint disease. These Mycoplasma species are distinct to the haemotropic mycoplasmas that target red blood cells, causing hemolytic anemia in cats. Mycoplasma felis is typically associated with Upper Respiratory Tract Disease (URTD) in cats. Transmission M. felis […]
Tritrichomonas Infection in Cat
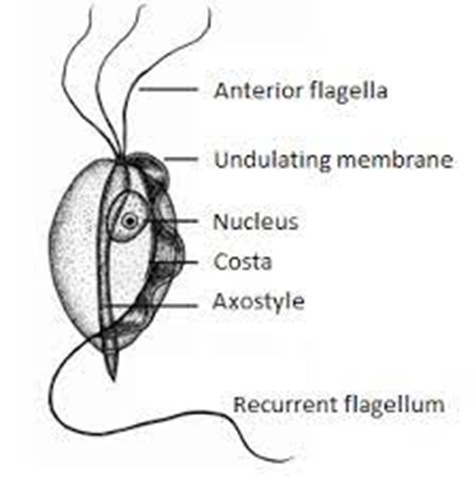
Bioguard Corporation Tritrichomonas foetus is a significant cause of large bowel diarrhea and persistent colitis in cats. These pear-shaped organisms have three anterior flagella and one posterior flagellum. They have a distinctive undulating membrane, which gives them a similar appearance to Giardia. However, they do not form cysts and are transmitted directly from one host […]
Encephalitozoon cuniculi in Rabbits

Bioguard Corporation Encephalitozoon cuniculi is a microsporidial, unicellular, spore-forming, obligate intracellular parasite. It can invade the host’s central nervous system, kidneys, crystals, etc. E. cuniculi affects rabbits by causing damage to the brain, nervous system, kidneys, and other important organs. E. cuniculi pose a zoonotic risk to immune-compromised humans. In addition, it can infect various […]
Psittacosis in Birds
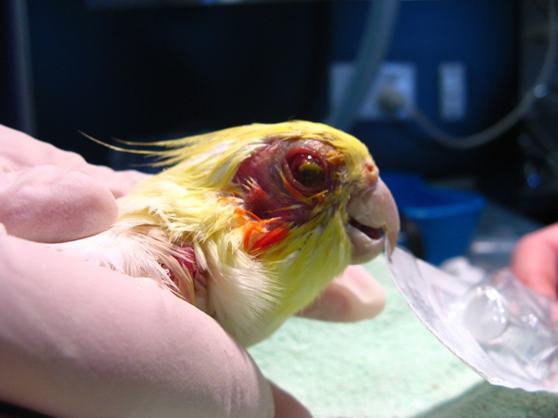
Bioguard Corporation Psittacosis (parrot fever), also known as ornithosis, is a bacterial zoonotic disease caused by the bacterium Chlamydia psittaci. It is usually spread by exposure to infected birds at home, pet stores, pigeon stalls and other locations where birds are kept or displayed. The disease was first reported in Switzerland in 1879, and then […]
Pigeon Circovirus

Bioguard Corporation Pigeon circovirus (PiCV) is an infectious disease that mainly affects young pigeons between 1 and 4 months of age. Mortality is variable, but it can approach 100%. Pigeon circovirus infections have led to the loss of lymphoid tissue in immune system organs, and for this reason, PiCV is regarded as an immunosuppressive agent […]
Lyme Disease in Dogs

Oliver Organista, LA Lyme disease is triggered by the bacterium Borrelia burgdorferi, which belongs to the spirochete class, characterized by its worm-like, spiral shape within the genus Borrelia. This bacterium is spread to both dogs and humans through the bite of an infected black legged tick, also known as the deer tick (Ixodes scapularis). The […]
Pancreatitis in Dogs

Dr. Sushant Sadotra Canine Pancreatitis is one of the most common endocrine diseases occurring in dogs. However, it is more prevalent in dog breeds such as Miniature Schnauzers, Yorkshire Terriers, Cocker Spaniels, Dachshunds, Poodles, and sled dogs. Pancreatitis, a severe inflammatory condition of the pancreas, can be short-term or long-term, based on the level of […]
Kidney Diseases in Cats

Lloyd Alexandria Chavez, R.M.T Cats possess a pair of kidneys located on either side of their abdomen, playing a crucial role in eliminating waste from their system. These organs are also key in regulating the balance of fluids, minerals, and electrolytes in the body, conserving water and protein, and supporting blood pressure and the […]
C-Reactive Protein
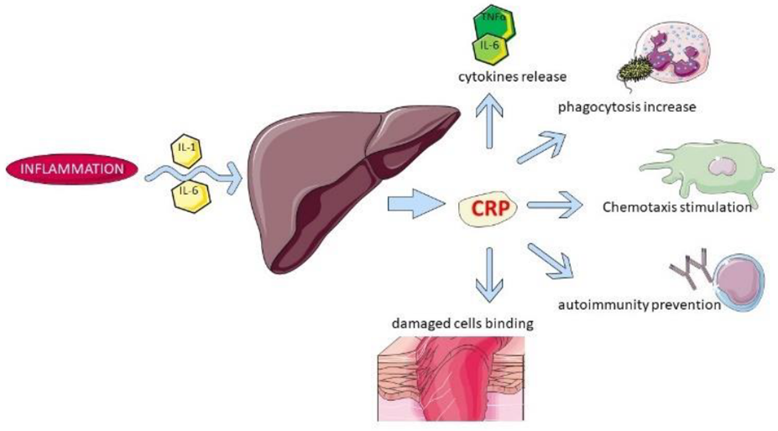
Sushant Sadotra The acute phase response (APR) is the early typical systemic response prompted by homeostasis disturbances such as injuries, infection, neoplasia, and other pathologies. Acute phase proteins get released from the liver into the blood after such stimulations. Therefore, elevated levels of such proteins can be used to indicate systemic inflammation. C-reactive protein, also […]
Respiratory Tract Disease Complex in Cats

Sushant Sadotra, PhD/Diagnostic specialist Feline respiratory disease (FRD) syndrome or feline upper respiratory tract disease complex is a common infection in cats caused mainly by Feline Herpesvirus (FHV-1), Feline Calicivirus (FCV), Chlamydophila felis, Mycoplasma spp., and Bordetella bronchiseptica. About 90% of all upper respiratory infections are caused by FHV-1 and FCV. Common Symptoms: · Sneezing · Nasal […]



