Feline Herpesvirus 1 (FHV-1) Infection

Bioguard Corporation Feline herpesvirus-1 (FHV-1) is a common viral infection in cats that primarily affects the upper respiratory system. It is a major cause of feline viral rhinotracheitis (FVR), which presents symptoms like sneezing, nasal discharge, and conjunctivitis (eye inflammation). FHV-1 is highly contagious among cats and is spread through direct contact with infected saliva, […]
Spinal Muscular Atrophy in Cats

Bioguard Corporation Spinal muscular atrophy (SMA) an autosomally recessive inherited neurodegenerative disorder seen in Maine Coon cats. The disease is characterized by weakness and atrophy in muscles due to loss of motor neurons that control muscle movement. Affected cats first show signs of disease around 3–4 months of age. Clinical signs include tremors, abnormal posture, […]
Polycystic Kidney Disease in Cats

Bioguard Corporation Polycystic kidney disease (PKD) is a chromosomally dominant genetic disorder; it can occur in humans, cats, dogs, and other animals. In the renal cortex and medulla, there are cysts of various sizes and fluid-filled, so it is commonly known as the bubble kidney. Cysts increase in size and number over time, replacing kidney […]
Feline Hypertrophic Cardiomyopathy
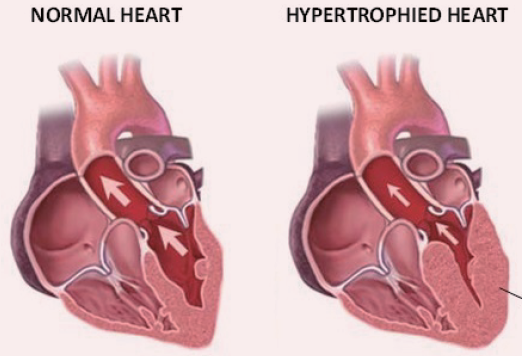
Bioguard Corporation Hypertrophic cardiomyopathy (HCM) is a primary, familial, and hereditary heart condition, and it is the most common heart disease in cats. Its key characteristic is primary concentric left ventricular hypertrophy (thickening of the heart wall), which occurs without pressure overload (such as from aortic stenosis), hormone stimulation (like in hyperthyroidism or acromegaly), myocardial […]
GM1 Gangliosidosis in Cats
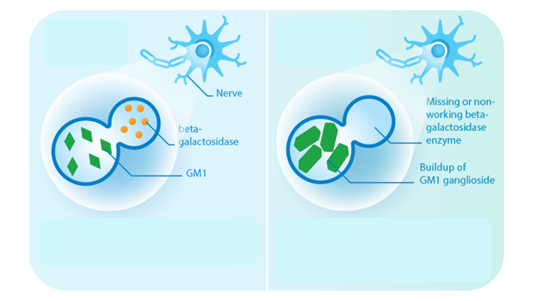
Bioguard Corporation GM1 gangliosidosis is a lysosomal storage disorder caused by deficiency of the enzyme β-galactosidase. Mutations in the GLB1 gene, encoding β-galactosidase, cause the progressive, neurosomatic, lysosomal storage disorder. Cats affected with gangliosidosis have progressive neurologic dysfunction around 3 months of age and premature death around one year old. Pathogenesis Gangliosides, normally hydrolyzed […]
Cryptosporidiosis

Bioguard Corporation Cryptosporidiosis is an illness you get from the parasite Cryptosporidium. It causes watery diarrhea and other gastrointestinal (gut) symptoms. In addition to stomach infection, this parasite can infect the respiratory system causing a cough and/or problems breathing. The family Cryptosporididae belongs to the phylum Apicomplexa characterized by an anterior (or apical) polar complex […]
Diagnosis of Feline Respiratory Mycoplasma Infection

Bioguard Corporation In cats, ’mucosal’ mycoplasma infections typically cause ocular and respiratory disease, and less frequently neurological or joint disease. These Mycoplasma species are distinct to the haemotropic mycoplasmas that target red blood cells, causing hemolytic anemia in cats. Mycoplasma felis is typically associated with Upper Respiratory Tract Disease (URTD) in cats. Transmission M. felis […]
Tritrichomonas Infection in Cat
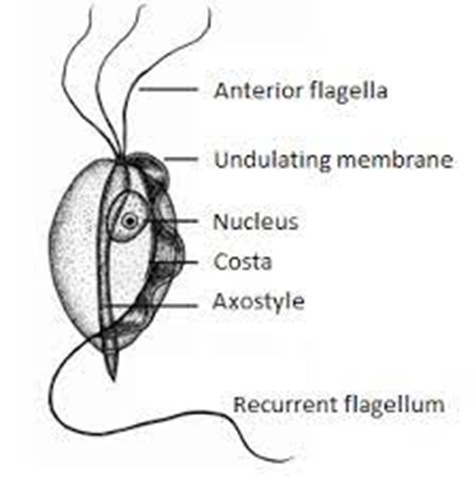
Bioguard Corporation Tritrichomonas foetus is a significant cause of large bowel diarrhea and persistent colitis in cats. These pear-shaped organisms have three anterior flagella and one posterior flagellum. They have a distinctive undulating membrane, which gives them a similar appearance to Giardia. However, they do not form cysts and are transmitted directly from one host […]



